Featured
A Mixture Of Nacl Sio2 And Caco3 Is Separated
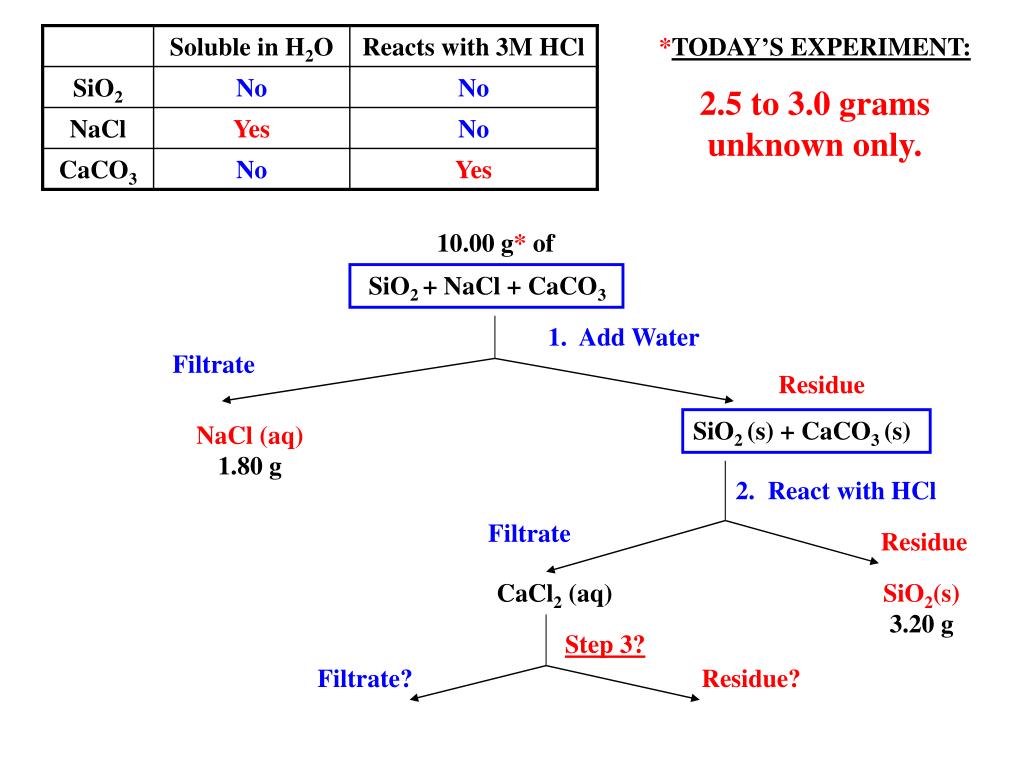
A Mixture Of Nacl Sio2 And Caco3 Is Separated. A mixture of nacl, sio2, and caco3 is separated following the procedure given in this experiment. Silicon dioxide and calcium carbonate can be separated based on a chemical property, reactivity with acid.
Use evaporation on the water and nacl beaker to isolate the nacl 5. “can you separate the mixture of sio3, caco3, and nacl by isolating the sio3 first or do you have to add water to separate the nacl first?” firstly, i’m assuming you mean sio_{2}, not sio_{3}. Indicate how each of the following procedural changes would affect the amount of the specified substance recovered, briefly explain the following reaction:
The Procedure Was To Seperate Nacl Caco3 And Sio2.
In my case, i used 10 ml hcl 37% to remove calcium carbonate from silica in pvc membrane filter. In this lab we are going to be given a mixture containing sodium chloride [nacl], silicon dioxide [sio2], iron filings [fe], and calcium carbonate [caco3] the goal of the lab is to use separation and measurement techniques to determine the percent by mass of each specie in the original sample. (2) the recovery of caco3 was.
The Calcium Carbonate So Obtainable Is Heated Strongly To Get 0.56G Of Cao.
This problem has been solved! The percentage of nacl in mixture is Weight of mixture before experiment 5.5380.
A Mixture Of Nacl, Sio2, And Caco3 Is Separated Following Theprocedure Given In This Experiment.
Indicate how each of the following procedural changes would affect the amount of the specified substance recovered. A mixture of nacl, sio2 and caco3 is separated following the procedure given in this experiment. You isolated 0.825 g nacl, 0.825 g of caco3.
The Recovery Of Caco3 Was Attempted By Adding 3M H2So4 Instead.
6hf + sio2 → h2sif6 + 2h2o identify the limiting reactant and excess reactant if 45 moles of sio2 and 140 moles of hf react. In the filter should be calcium carbonate and in. Science chemistry q&a library a mixture of nacl, caco3, and sio2 is separated using the techniques in the procedure of this lab.
Indicate How Each Of The Following Procedural Changes Would Affect The Amount Of The Specified Substance Recovered.
In order to recover the nacl as a solid, we simply boil the water. (atomic mass of ca = 40) a. Could the separation in this experiment have been done in a different order?
Popular Posts
Difference Between Pure Substance And A Mixture
- Get link
- X
- Other Apps
Comments
Post a Comment